Karenia mikimotoi (Miyake & Kominami ex Oda) Gert Hansen & Moestrup 2000
Current name:
Karenia mikimotoi (Miyake & Kominami ex Oda) Gert Hansen & Moestrup
Ireland.Caroline Cusack (robin.raine@nuigalway.ie)
Publication Details
Karenia mikimotoi (Miyake & Kominami ex Oda) Gert Hansen & Moestrup 2000: 308
Published in: Daugbjerg, N., Hansen, G., Larsen, J. & Moestrup, Ø. (2000). Phylogeny of some of the major genera of dinoflagellates based on ultrastructure and partial LSU rDNA sequence data, including the erection of three new genera of unarmoured dinoflagellates. Phycologia 39: 302-317.
Type Species
The type species (holotype) of the genus Karenia is Karenia brevis (C.C.Davis) Gert Hansen & Moestrup.
Status of Name
This name is of an entity that is currently accepted taxonomically.
Basionym
Gymnodinium mikimotoi Miyake & Kominami ex Oda
Type Information
Type locality: NW Pacific Ocean: Gokasho Bay, Japan; (Faust & Gulledge 2002: 40) Holotype: (Faust & Gulledge 2002: 40)
General Environment
This is a marine species.
Description
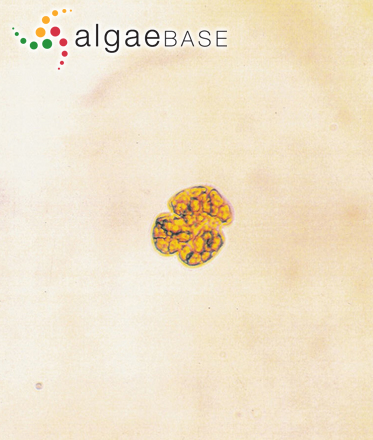
A photosynthetic species with several oval to round yellow-brown chloroplasts, each with a pyrenoid. The large ellipsoidal nucleus is located in the left hypothecal lobe. No thecal plates are present. Cells are small, broadly oval to almost round and compressed dorso-ventrally. Cells are slightly longer than wide with a characteristic long and straight apical groove to the right of the sulcal axis. The apical groove extends from the ventral side to the dorsal side of the epitheca creating a slight indentation at the apex of the cell. Cells range in size from 18-40 µm in length to 14-35 µm in width .The epitheca is broadly rounded and smaller than the hypotheca. The hypotheca is notched by the widening sulcus at the antapex resulting in a lobed posterior. The wide and deeply excavated cingulum is pre-median, and is displaced in a descending spiral about 2 times its width. The sulcus slightly invades the epitheca extending from above the cingulum to the antapex (adapted from http://www.nmnh.si.edu/botany/projects/dinoflag/Taxa/Gmikimotoi.htm).
Habitat
A planktonic species first described from western Japan. This species is a recurring bloom former in coastal waters of Japan and Korea; red tides commonly occur in warmer months and are associated with massive fish and shellfish kills. Reported to be eurythermal and euryhaline, populations of G. mikimotoi could presumably over-winter as motile cells, which could then serve as seed populations for a summer red tide. Moreover, studies conducted in Omura Bay, Japan, revealed that this species can tolerate anoxic or near anoxic conditions utilizing sulfide from the sediment.
Key Characteristics
Cells have a distinct swimming pattern: turning over through water, like a falling leaf.
Created: 26 April 2002 by M.D. Guiry.
Last updated: 03 August 2023
Verification of Data
Users are responsible for verifying the accuracy of information before use, as noted on the website Content page.
Linking to this page: https://www.algaebase.org/search/species/detail/?species_id=44334
Citing AlgaeBase
Cite this record as:
G.M. Guiry in Guiry, M.D. & Guiry, G.M. 03 August 2023. AlgaeBase. World-wide electronic publication, National University of Ireland, Galway. https://www.algaebase.org; searched on 19 April 2024
 Request PDF
Request PDF














